Key Labeling Requirements for U.S. Dietary
Supplements under 21 CFR
Introduction
Dietary supplements sold in the United States must comply with labeling requirements established by the U.S. Food and Drug Administration (FDA). These requirements are primarily defined under 21 CFR Part 101, which specifies how supplement labels must present ingredient information, nutrition facts, and regulatory statements.
Among these regulations, 21 CFR §101.36 specifically describes how the Supplement Facts panel should be structured and how dietary ingredients must be declared.
Official regulation reference:
https://www.ecfr.gov/current/title-21/section-101.36
The following section summarizes the key labeling elements that must appear on a dietary supplement product sold in the U.S. market.
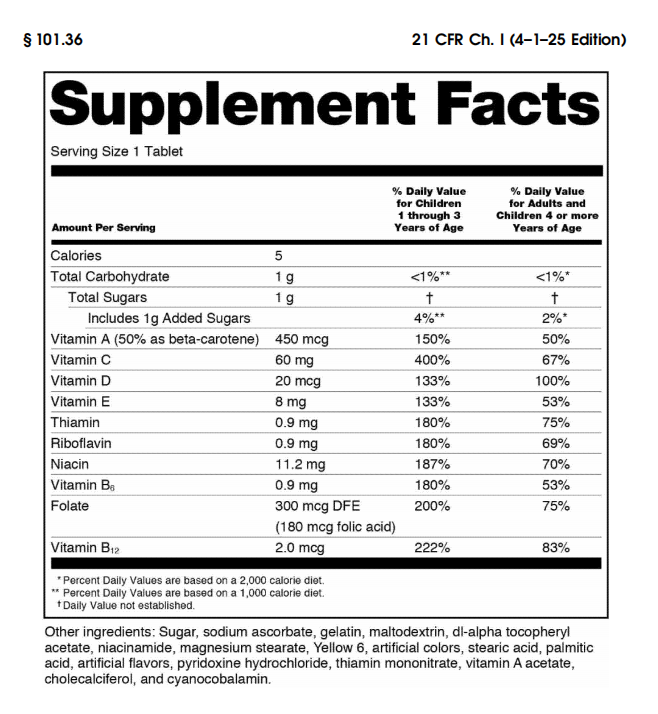
Core Labeling Requirements for Dietary Supplements
Under U.S. regulations, dietary supplement labels must include several mandatory elements. These elements ensure that consumers receive transparent and standardized product information.
1. Statement of Identity
The label must clearly identify the product as a dietary supplement.
Examples:
- Vitamin D Dietary Supplement
- Magnesium Dietary Supplement
- Herbal Dietary Supplement
This statement must appear on the principal display panel (PDP) of the packaging.
2. Net Quantity of Contents
The label must indicate the total amount of product contained in the package.
Examples include:
- 60 Capsules
- 120 Tablets
- Net Weight 150 g
This information must be displayed clearly on the front label panel.
3. Supplement Facts Panel
The Supplement Facts panel is the most important part of dietary supplement labeling.
According to 21 CFR §101.36, the panel must include the following elements:
• Serving Size
• Servings Per Container
• Name of each dietary ingredient
• Amount of each ingredient per serving
• Percent Daily Value (%DV), when applicable
The heading “Supplement Facts” must appear prominently at the top of the box.
The panel must also follow standardized formatting rules, including typography, indentation, and boxed layout.
4. Declaration of Dietary Ingredients
Each dietary ingredient must be declared with its quantitative amount per serving.
For example:
Vitamin C (as ascorbic acid) — 500 mg
Magnesium (as magnesium citrate) — 200 mg
The declared amount refers to the actual amount of the nutrient or dietary ingredient, not the total weight of the raw material.
For example:
Magnesium citrate providing 200 mg elemental magnesium must be labeled as:
Magnesium — 200 mg
rather than listing the raw material weight.
5. Botanical Ingredient Identification
For botanical ingredients, the label must specify the plant part used.
Examples:
Milk Thistle Extract (seed)
Turmeric Extract (root)
Ginkgo Biloba Extract (leaf)
This helps ensure transparency regarding the botanical source.
6. Other Ingredients List
Ingredients that are not dietary ingredients must be listed under “Other Ingredients.”
Examples include:
• Capsule shell materials
• Flow agents
• Fillers
• Stabilizers
These ingredients must be listed in descending order by weight.
7. Name and Address of Responsible Party
The label must identify the manufacturer, packer, or distributor responsible for the product.
Typical format:
Manufactured for
Company Name
City, State, ZIP Code
Country (if imported)
This requirement allows traceability of the product.
8. Structure / Function Claims and Disclaimer
Dietary supplements may include structure/function claims, such as:
- Supports immune health
- Helps maintain bone health
- Supports normal energy metabolism
However, these claims must not imply disease treatment.
When such claims are used, the following disclaimer must appear on the label:
“These statements have not been evaluated by the Food and Drug Administration.
This product is not intended to diagnose, treat, cure, or prevent any disease.”
This requirement originates from the Dietary Supplement Health and Education Act.
Common Labeling Compliance Risks
Companies entering the U.S. supplement market often encounter labeling issues due to misunderstanding regulatory requirements. The following are some common compliance risks.
Incorrect declaration of nutrient dosage
One of the most common mistakes is declaring the raw material weight instead of the actual nutrient amount.
Example:
Incorrect:
Magnesium Citrate — 1000 mg
Correct:
Magnesium — 200 mg (from Magnesium Citrate)
FDA labeling requires declaration of the nutrient amount, not simply the ingredient weight.
Missing plant part for botanical ingredients
Botanical ingredients must specify the plant part used.
Incorrect example:
Milk Thistle Extract — 300 mg
Correct example:
Milk Thistle Extract (seed) — 300 mg
Failure to identify the plant part may lead to labeling compliance issues.
Incorrect Supplement Facts formatting
The Supplement Facts panel must follow specific layout and formatting rules.
Common issues include:
• incorrect indentation
• missing serving size information
• incorrect placement of % Daily Value
• improper font hierarchy
Even small formatting errors may lead to regulatory concerns.
Unapproved disease claims
Supplements cannot claim to diagnose, treat, cure, or prevent diseases.
Examples of problematic claims include:
- Treats arthritis
- Prevents diabetes
- Cures liver disease
Such statements would classify the product as a drug under FDA regulations.
Missing FDA disclaimer
When structure/function claims appear on the label, the DSHEA disclaimer must be included.
Failure to include this statement may result in regulatory violations.
Regulatory References
For detailed regulatory language, the following official references can be consulted.
21 CFR §101.36 – Nutrition labeling of dietary supplements
https://www.ecfr.gov/current/title-21/section-101.36
FDA Dietary Supplement Labeling Guide
https://www.fda.gov/food/dietary-supplements-guidance-documents-regulatory-information/dietary-supplement-labeling-guide
Electronic Code of Federal Regulations
https://www.ecfr.gov/current/title-21
Conclusion
Understanding the labeling requirements defined under 21 CFR is essential for companies developing or exporting dietary supplements to the United States.
Accurate ingredient declaration, correct Supplement Facts formatting, and compliant structure/function claims are key elements of regulatory compliance. Careful attention to these requirements can help reduce market entry risks and ensure smoother product commercialization.
Synergistic Effects of Creatine and
HMB in Sports Nutrition
Introduction
Creatine and β‑Hydroxy β‑methylbutyrate (HMB) are two widely used ingredients in sports nutrition products.
Creatine mainly improves muscle energy availability, while HMB primarily reduces muscle protein breakdown. Because their mechanisms differ, researchers have studied whether combining them can enhance training outcomes.
A controlled resistance-training study published in the Journal of Strength and Conditioning Research reported improved strength and lean body mass when creatine and HMB were supplemented together.
Study reference
Authors: William J. Kraemer et al.
Publication year: 2009
Original article
https://journals.lww.com/nsca-jscr/Abstract/2009/05000/The_Effects_of_Creatine_and_HMB_Supplementation.1.aspx

Key Mechanisms
Creatine – Energy Production
Creatine increases phosphocreatine stores in skeletal muscle, which helps regenerate ATP during short-duration high-intensity exercise.
Main benefits observed in studies:
• improved maximal strength
• increased training volume
• improved sprint or power performance
HMB – Muscle Protein Protection
HMB is a metabolite of leucine that helps reduce muscle protein breakdown during intense training.
Reported effects include:
• reduced exercise-induced muscle damage
• improved recovery
• preservation of lean muscle mass
The most common commercial form used in supplements is Calcium HMB (HMB-Ca).
Evidence of Combined Supplementation
Research combining creatine and HMB supplementation during resistance training has reported:
• greater increases in lean body mass
• improved maximal strength
• reduced markers of muscle damage
Because creatine enhances energy availability while HMB supports muscle protein balance, the combination may provide complementary benefits for strength training programs.
Common dosage ranges used in sports nutrition products:
Creatine Monohydrate
3–5 g per serving
Both ingredients are commonly used in:
• sports performance powders
• pre-workout formulas
• muscle recovery blends
Conclusion
Creatine and HMB-Ca are frequently combined in sports nutrition products due to their complementary physiological roles. Research suggests that this combination may support improved strength gains and lean mass development during resistance training.
